Intradermal injections of a hair growth factor formulation for enhancement of human hair regrowth – safety and efficacy evaluation in a first-in-man pilot clinical study
QR678 NEO: NATURAL INGREDIENTS
BACKED BY SCIENCE
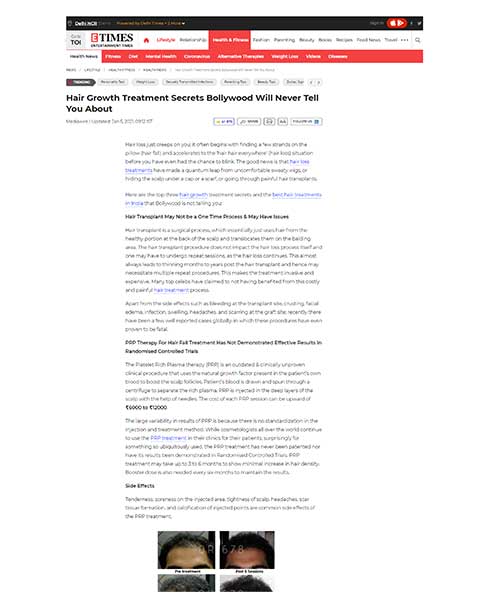
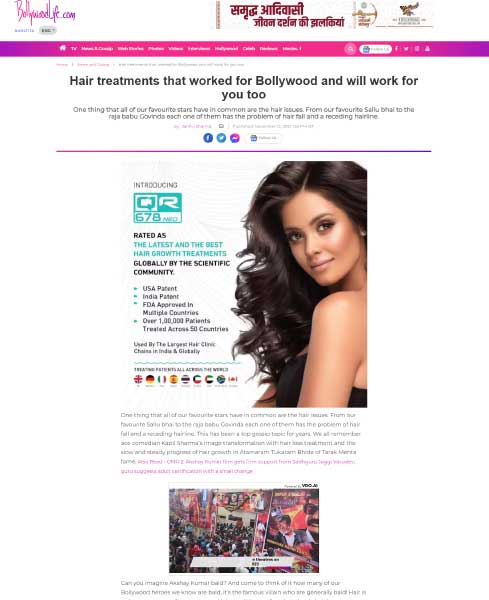
The QR678® Neo therapy is a proprietary, first-in-class therapy, backed by science that arrests hair fall & increases the thickness, number, and density of existing hair follicles leading to greater coverage in hair loss. The biomimetic polypeptides used in the formulation mimic the action of specific human hair growth related growth factors that are naturally present in our scalp, making it natural growth factor-based treatment. The polypeptides penetrate deep into the scalp and provide nourishment to the scalp, which results in hair growth. Unlike other treatments that contains minoxidil & finasteride, QR678® Neo therapy is completely devoid of side effects.
HOW DOES IT WORK?
QR678® Neo ADVANTAGES

(Derived From Plants)

Quick & Effective (Single
Session in 10 Minutes)

Non Surgical

Success Rate
13+ Published Researched Clinical Trials Demonstrating Efficacy
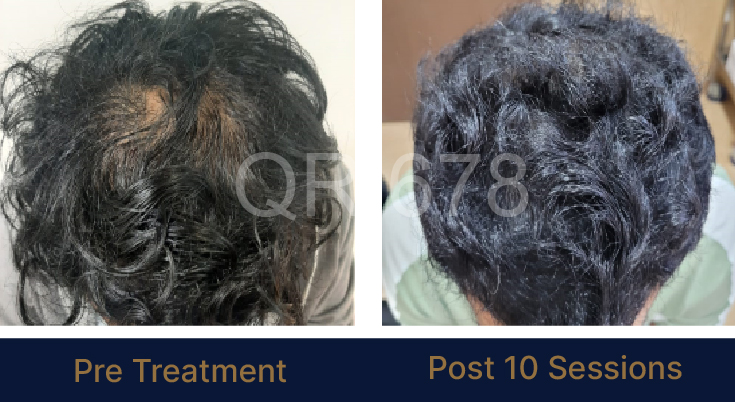
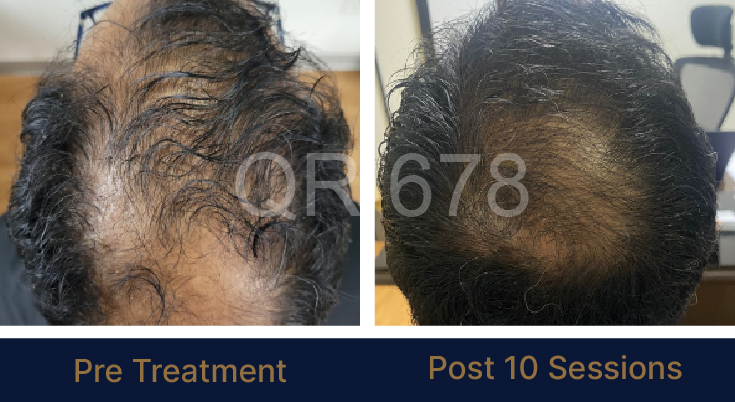
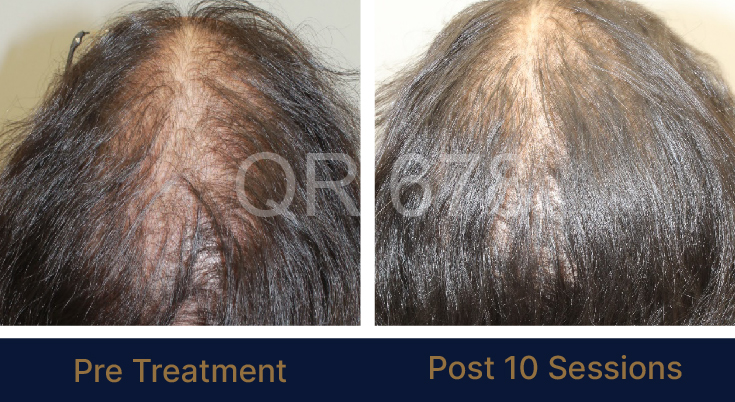
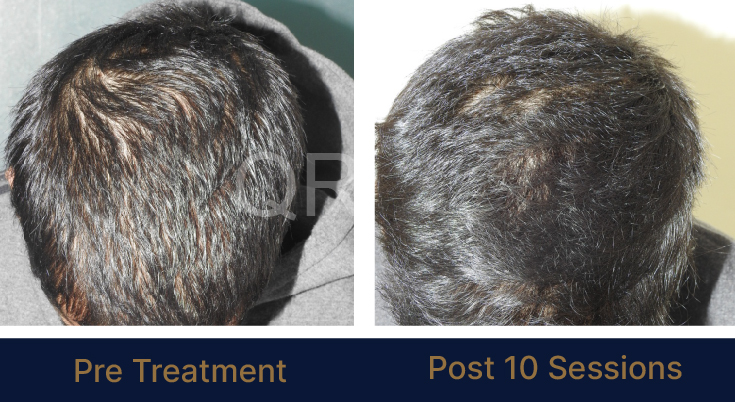
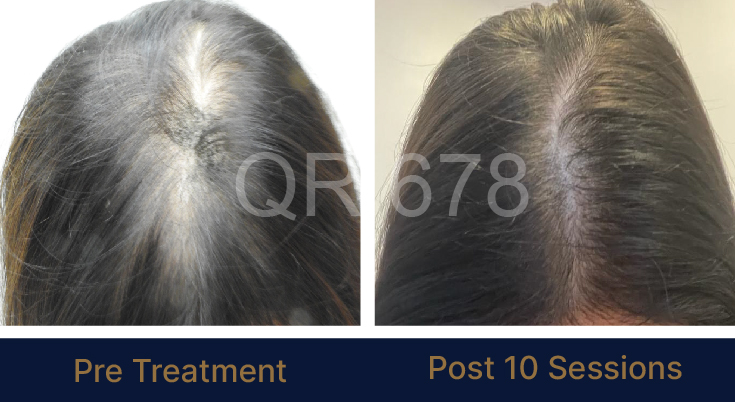
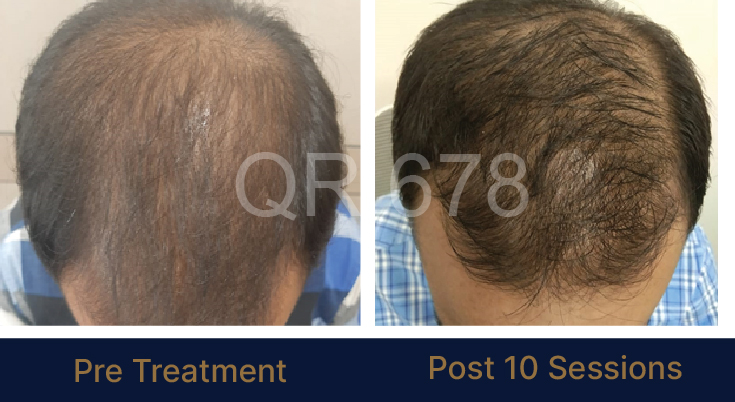
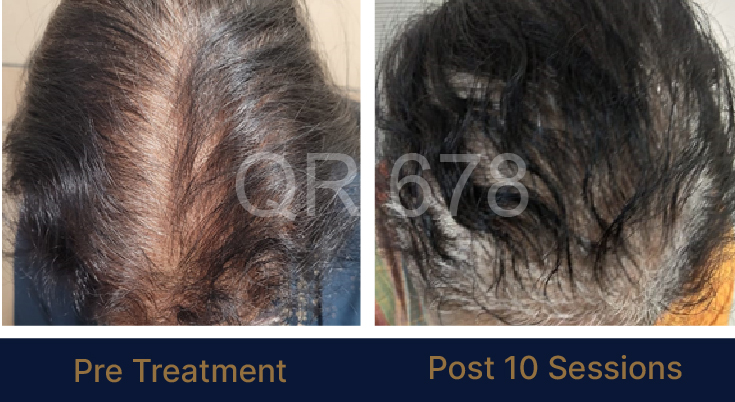
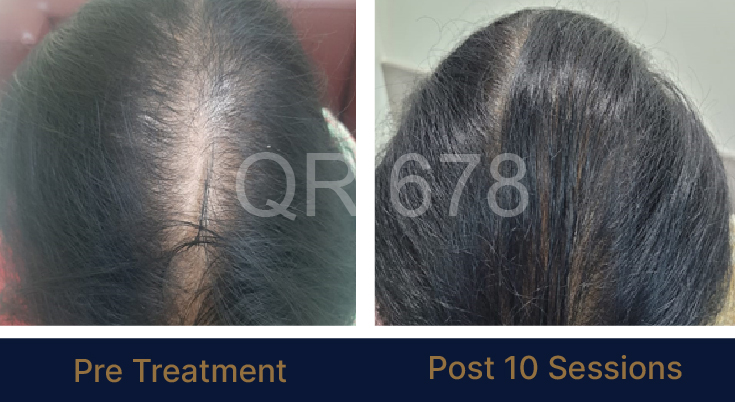
LIFE CHANGING RESULTS
PRE TREATMENT
POST 6 SESSIONS
PRE TREATMENT
POST 6 SESSIONS

PRE TREATMENT
POST 6 SESSIONS
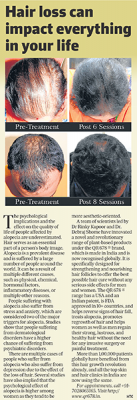
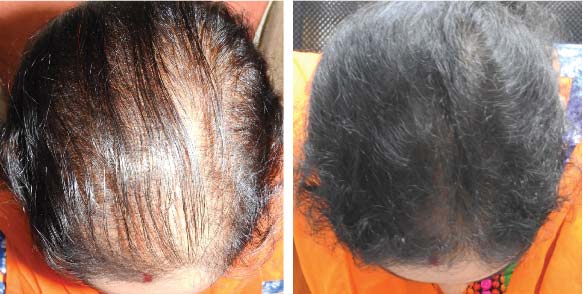
25 year old female from alopecia areata, was also diagnosed with hypothyroid. She achieved impressive results with QR678, a completely steroid-off treatment.

PRE TREATMENT
POST 6 SESSIONS
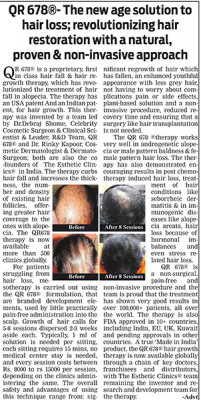
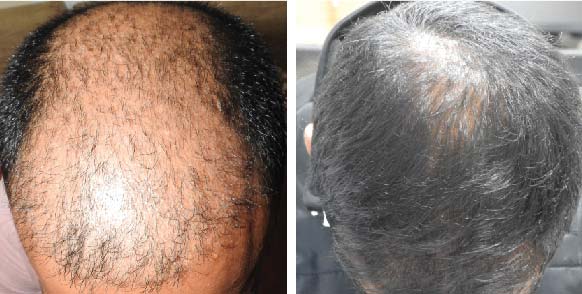
39 year old male suffering from male pattern hair loss for 15 years gained a significant amount of hair with QR678
MEDIA INTERVIEWS
TESTIMONIALS
HIGHLIGHTS IN MEDICAL CONFERENCES





CUTICON – December 2023









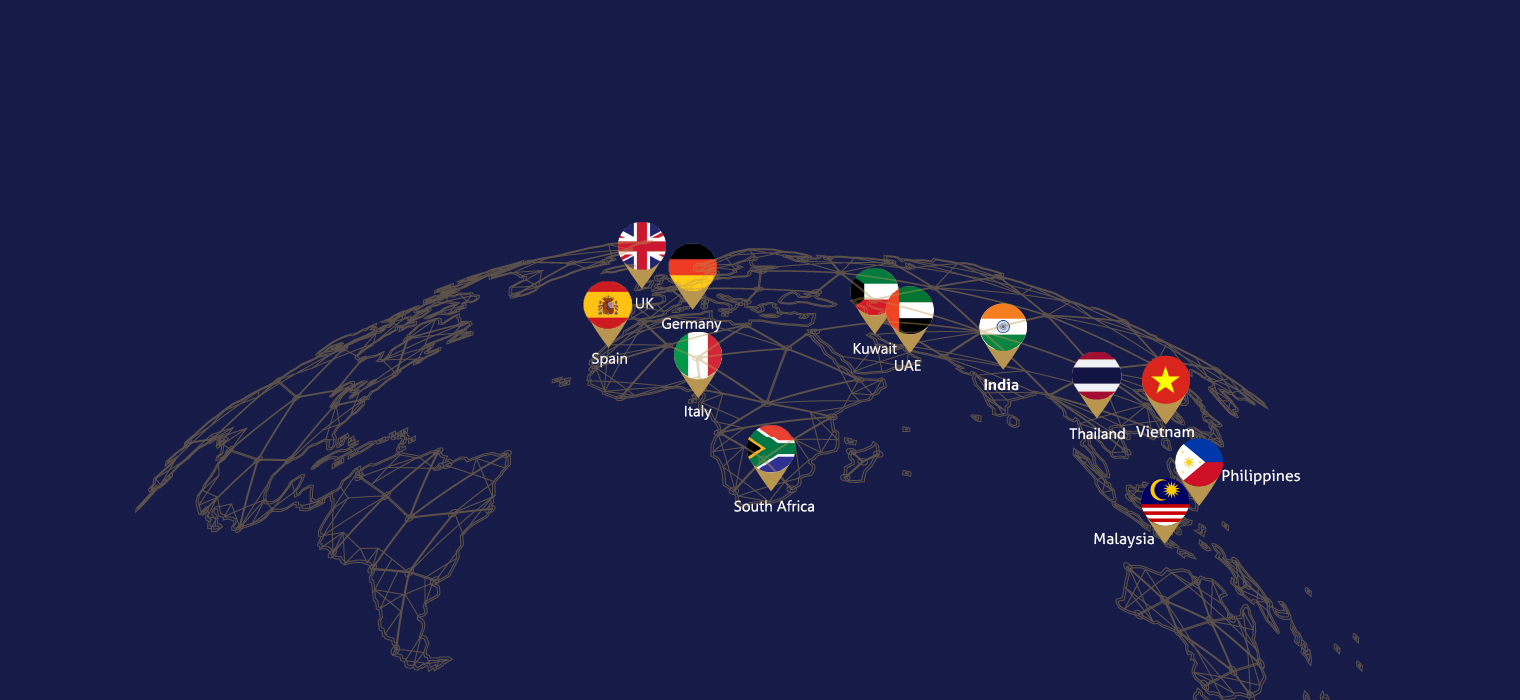
GLOBAL PRESENCE
OUR PARTNERS

















FAQ'S
How is QR678® Neo better than other hair fall treatments?
QR678® Neo is a mixture of natural growth factors that are already present in the scalp and therefore it is completely safe to use on everybody. It has the right concentration of specific hair growth factors and QR678® Neo does not have any side effects, is performed as an outpatient procedure and the results are visible within eight weeks of starting the treatment. You can listen for yourself what the patients say about QR678® Neo hair fall treatment https://www.youtube.com/watch?v=pIvjpQgSsrc
How often do I need to repeat QR678® Neo treatment?
QR678® Neo injections are administered in 8-12 sessions in about 3-4 weeks apart. The results are visible after few weeks. Once you have achieved the desired results the injections need not be repeated unless the hair fall starts again. This is because QR678® Neo make the hair follicles healthy. Get to know more about why QR678® Neo is highly effective in treating hair fall and increasing hair growth. http://www.qr678.in/
What precautions should I take after QR678® Neo treatment?
QR678® Neo is a completely safe treatment without side effects and you can go about your daily routine after the sessions. The only care you need to take is to use a gentle shampoo not before the next day of the treatment and avoid scratching and rubbing the treating area. If you have any other problem then consult your dermatologist. https://www.youtube.com/watch?v=LNm2z3EGLxg
Can QR678® Neo be used after hair transplant?
Yes, using QR678® Neo after hair transplant produces very successful results. QR678® Neo gives faster density and reduces the catagen loss of transplanted hair. The growth factors in QR678® Neo help in faster healing and activate the dormant follicles and thus - Increase the viability of hair follicles - Improve healing and repairing of tissue process - Strengthen the inactive hair follicles and stimulate hair growth - Improves the health of existing hair and they will become fuller and stronger QR678® Neo can be given in multiple treatment sessions after three to four weeks after successful hair transplant surgery. QR678® Neo can also be used before the hair transplant surgery to reduce the area of transplant and improve the results.
How is QR678® Neo better than PRP and stem cell hair fall treatment?
QR678® Neo is a relatively new treatment for hair fall and hair regrowth but in the short time of its invention QR678® Neo has proven to be more effective than PRP and stem cell treatment for reducing hair fall. QR678® Neo produces longer lasting and better results and is completely safe and without any side effects. QR678® Neo can help treat and rogenetic alopecia, female pattern hair loss, and hair loss because of chemotherapy, seborrheic dermatitis, and alopecia areata. PRP and stem cell on the other hand do not have the specific concentration of required growth factors, they lack standardisation in terms of treatment protocols and preparation of the solution injected and lack good quality studies to prove their benefits. produce guaranteed results and can cause tightness and tenderness in scalp, scar tissue formation, calcification of the injected points, and formation of scar tissues. Both processes are still under investigation and their results are still awaiting confirmation. Dr Rinky Kapoor of The Esthetic Clinics explains the difference between QR678® Neo and stem cell here: https://www.youtube.com/watch?v=wgNihm8ciu4 and https://www.youtube.com/watch?v=ZUWXKzVh0PM
Can genetic hair loss be treated?
Yes genetic hair loss can be treated if the treatment is started in time. Heredity hair loss usually runs in the family and can affect both men and women. If you too have a close elder relative who lost his or hair in a patterned baldness then you should keep a close eye on your hair too. Hereditary hair loss usually happens in a pattern; M or W shaped in men and along the hair part in women. See the doctor immediately if you suffer from hair loss. There are many common and successful treatments available for genetic hair loss which include
- Topical and oral medications: These include Minoxidil topical lotion and Finasteride tablets.
- Hair transplantation using FUT and FUE methods.
- Nutritional supplements: Your physician might also suggest some hair supplements that can help arrest hair fall. Dietary supplements containing Vitamin A, C, and Biotin, zinc, and iron can also help control hair fall caused because of genetics.
Treatment option for Chemotherapy induced Alopecia?
Alopecia post chemotherapy is a very common consequence of cancer treatments and therapies and it is often the most psychologically and socially devastating side effect. Many patients shy away from the treatments because of fear of hair fall and hair loss. However, in most cases, such alopecia is reversible but the recovery takes several months and even up to a year. Some basic latest treatment methods will speed up the healing: - Scalp Cooling: This is a FDA approved method for patients suffering from breast cancer. Scalp cooling or hypothermia results in constriction of the blood vessels in the scalp, which in turn leads to reduced delivery of chemotherapy to the hair. Scalp cooling devices consist of cooling unit that circulate the coolant on the scalp. This is done about 30 minutes before the start of chemotherapy. Patients can feel some mild discomfort, nausea, and dry skin because of scalp cooling. - Topical Minoxidil: Minoxidil helps by increasing the blood flow to the hair follicle thereby preventing miniaturization of hair follicle. - QR678® Neo hair regrowth treatment: QR678® Neo is a FDA approved, completely safe hair regrowth treatment that has no side effects or causes any discomfort to the patient. Clinical trials have proved QR678 to be effective in treatment of chemotherapy induced alopecia in both men and women. The results of QR678® Neo last lifelong.
FOR MORE QUERIES
Country: India Bangladesh Kuwait Thailand UAE