Invention of QR678® platform technology:
The QR678 research & development platform technology is a proprietary first-in-class technology. This science backed technology is giving rise to multiple products that arrest hair fall and address the thickness, density of hair follicles leading to greater coverage in hair loss. This R&D technology deals with multiple human hair growth related growth factors that are naturally present in our scalp making products derived from these completely natural. Research on this platform technology has resulted in this being awarded USA & Indian patents and has led to over 16+ clinical research papers being published with products derived from this unique technology. The hair growth factors penetrate deep into the scalp and provide nourishment to the scale. Unlike other treatments that contains harmful medication, products derived from QR678 are almost completely devoid of side effects.
QR678®, a novel research and development technology, patented in the USA and India & draws on years of research and scientific validations to create a unique formulation of hair growth factors along with various vitamins, minerals and amino acids.
The QR678® technology is being used to develop multiple products. Since this technology is very novel and patented, it is estimated that these products would be used by millions to address issues related to hair loss.
We call it
Researched. Recognised. Revolutionary.
For a reason, or many.
Researched:
- Developed after rigorous research for over a decade
- 16+ research papers published in top international journals
- Being tried and tested for multiple indications
Recognised:
- USA and India patent
- Products derived from this have regulatory approval in 20+ countries
- Used by 1500+ doctors and clinics globally
Revolutionary:
- First-in-line solution for hair loss
- Assisted over 150,000 patients worldwide
How does it work?
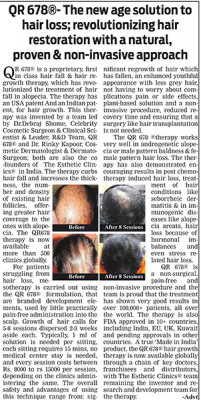
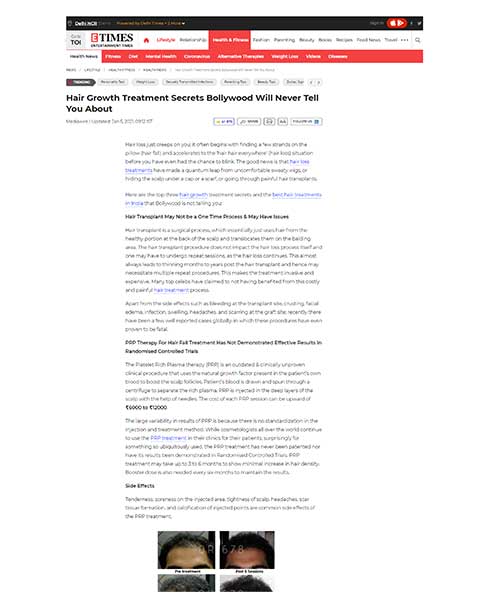
The QR678® therapy arrests hair fall & increases the thickness, number & density of existing hair follicles, leading to greater coverage in patients with alopecia. Since, the growth factors used in QR678® are anyways present in scalp full of hair (they get decreased in scalps which have hair fall), it is the enrichment of the scalp skin with these growth factors which cause hair growth. Since these growth factors are anyways normally present in the scalp, replenishing the scalp with these is not artificial and does not have any side effects.
The QR678® therapy works very well in androgenetic alopecia or male pattern baldness & female pattern hair loss. The QR678® therapy has also demonstrated encouraging results in post chemotherapy induced hair loss, seborrheic dermatitis induced hair loss & in immunogenic diseases like alopecia areata.
In our new products, the team has formulated a blend of bio-mimicking polypeptides (analogous to naturally occurring human protein factors) with amino acids and minerals to produce astonishing results, compared to Platelet Rich Plasma (PRP) based therapy or Stem cells therapy or even in cases otherwise refractory to Minoxidil or Finasteride or a combination therapy. This treatment works very well in male and female pattern baldness, and even in post hair transplant cases. In early cases of hair loss (Norwood Grade I – III), this treatment is actually showing better results than hair transplant surgery.
How was it invented & Invention Timelines:
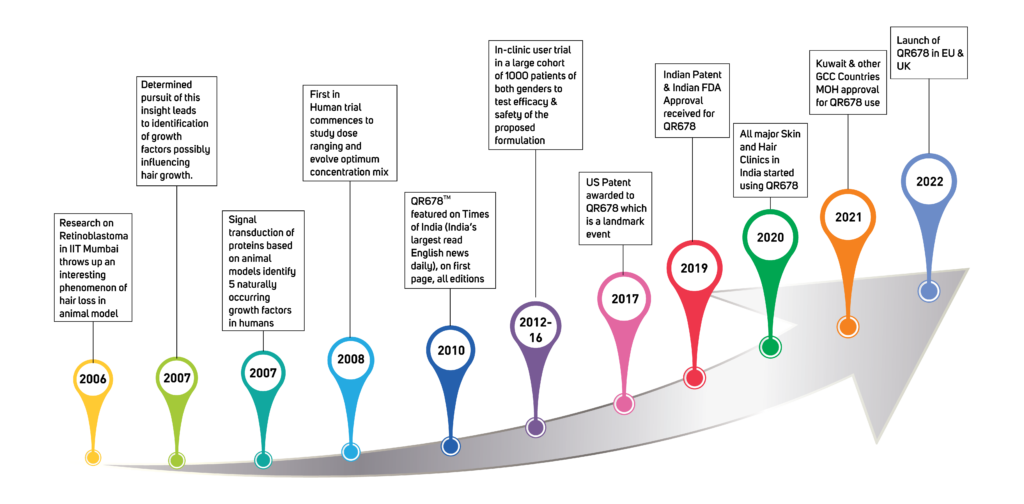
In 2006, While working on a drug for ocular & neural cancers, Dr. Shome stumbled upon potential causes of hair fall, in the animal trial he was pursuing.
This research on Retinoblastoma conducted in collaboration with the Indian Institute of Technology, Mumbai, India (IIT) threw up an interesting phenomenon of hair loss in the animal model.
From 2006-2008, Determined pursuit of this insight leads to identification of growth factors and polypeptides possibly influencing hair growth. Signal transduction of proteins, based on animal models, were used to identify 5 naturally occurring growth factors/ polypeptides in humans.
In 2009, the First in Human trial commenced to study dose ranging and evolve optimum concentration mix to yield the best in class results.
The QR678® Neo hair formulation therapy was featured on Times Of India (India’s largest read English news daily), on first page, all editions on September 19th, 2010.
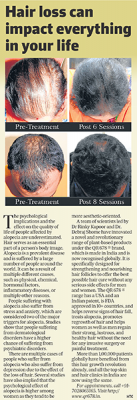
Subsequently, from 2013-2016, a Phase III clinical user trial in a large cohort of 1000 patients of both genders was completed, to test efficacy & safety of the proposed formulation. This clinical trial was published in a very reputed, top, American Journal, Journal of Cosmetic & Laser Therapy, in 2017. After 8 sessions, global photographs showed improvement from baseline for 71% patients, a decreased score for 10% patients, & no change in score for 19% patients. The findings of this study suggest that the beneficial clinical effects of this QR 678 ® hair formulation therapy are similar in men & women, across different age groups, & in patients irrespective of the presence of metabolic disorders like diabetes, hypertension, hypercholesterolemia etc.
Assessed In Clinical Trials
QR678® has been through a prospective, single arm interventional CLINICAL Trial, in which 1000 patients were given administration into the scalp skin. The treatment regimen comprised 8 sessions, 3 to 4 weeks apart, till 8 sessions were completed. Hair pull test was performed, before every session. Videomicroscopic & global images were taken at baseline, fourth session, eighth session, & 2 months after the completion of the 8 sessions. Relevant safety assessments through physical examination, questionnaires & appropriate laboratory examination were conducted throughout the study.
The treatment was effective in improving the appearance of scalp hair & slowing the loss of hair in men & women with patterned hair loss. Improvement in hair growth with therapy was evident as early as after 4 sessions for all measured endpoints. At one year, a statistically significant increase in total hair count (P=0.002) continued to be seen. After 8 sessions, global photographs showed improvement from baseline for 71% patients, a decreased score for 10% patients, & no change in score for 19% patients.
The findings of this study suggest that the beneficial clinical effects of this therapy are similar in men & women, across different age groups, & in patients irrespective of the presence of metabolic disorders like diabetes, hypertension, hypercholesterolemia etc. Moreover, results indicate that therapy was also effective in controlling hair loss in 14 post hair transplant patients. There was a negative correlation between the duration & stage of hair loss, & the degree of improvement.
Haematology & serum biochemical analysis were performed at baseline, sessions 4, 8 & then 2 months post the 8th session. There was no significant change in the biochemical values from the administration of QR678®.
Intellectual Property Assets:
A composition & mechanism of administration patent was awarded to the QR678® research and development hair science platform on 11th July 2017, by the United Stated Patent & Trademark Office (USPTO) – USA patent no 9, 700, 504 B2. Details of the patent are available below:
The Indian Patent Application was granted a composition and method of administration comprehensive patent, vide Indian Patent No. IN310925, granted on 5th April, 2019.
QR678®: SCIENCE Backed Research & Development Proprietary Product Technology
The QR678® research & development platform technology is a proprietary first-in-class technology. This science backed technology is giving rise to multiple products that arrest hair fall and address the thickness, density of hair follicles leading to greater coverage in hair loss. This R&D technology deals with multiple human hair growth related growth factors that are naturally present in our scalp making products derived from these completely natural. Research on this platform technology has resulted in this being awarded USA & Indian patents and has led to over 16+ clinical research papers being published with products derived from this unique technology. The hair growth factors penetrate deep into the scalp and provide nourishment to the scale. Unlike other treatments that contains harmful medication, products derived from QR678® are almost completely devoid of side effects.
HOW DOES IT WORK?
QR678® ADVANTAGES

(Derived From Plants)

Quick & Effective (Single
Session in 10 Minutes)


Non Surgical

Success Rate
13+ Published Researched Clinical Trials Demonstrating Efficacy
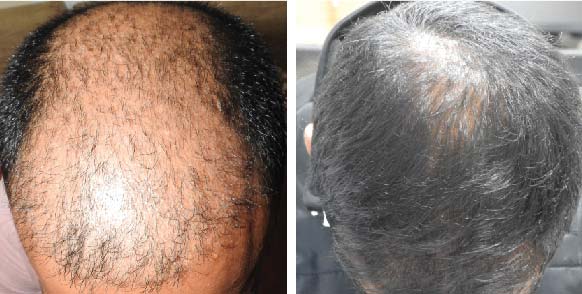
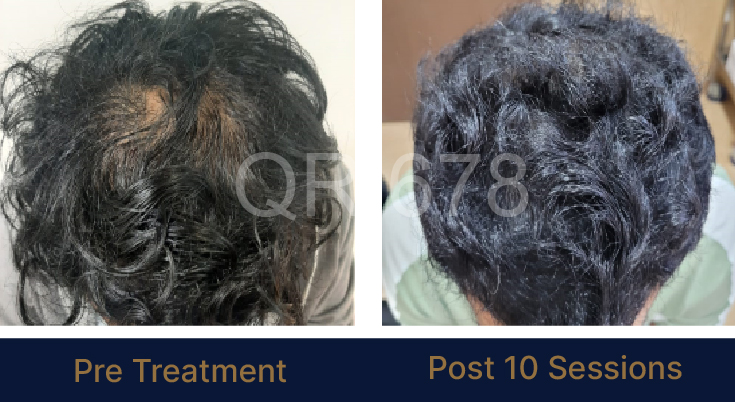
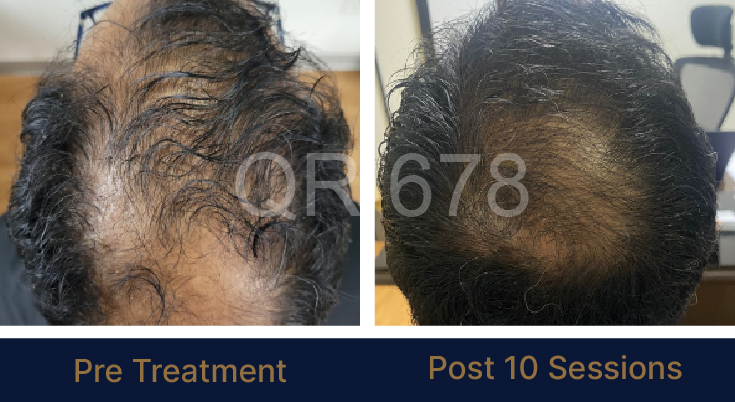
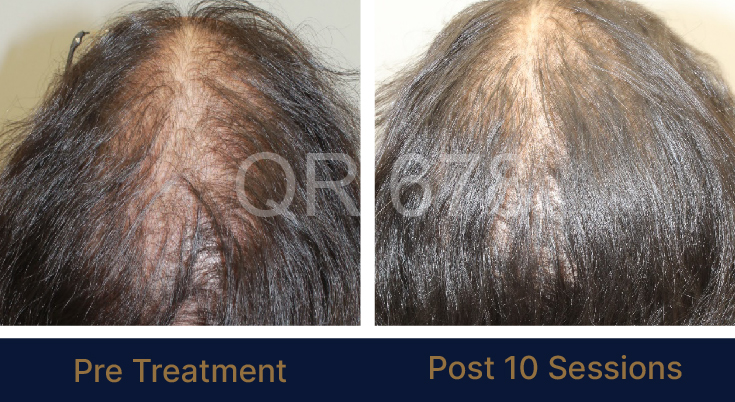
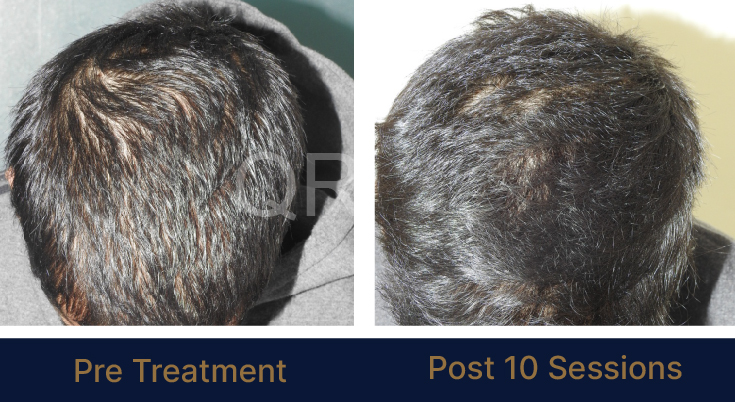
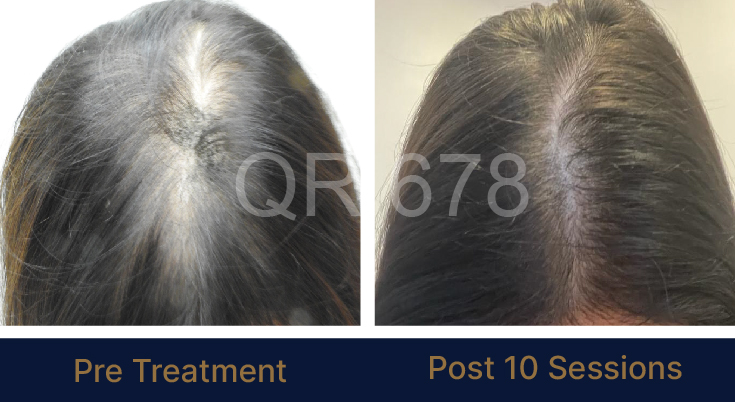
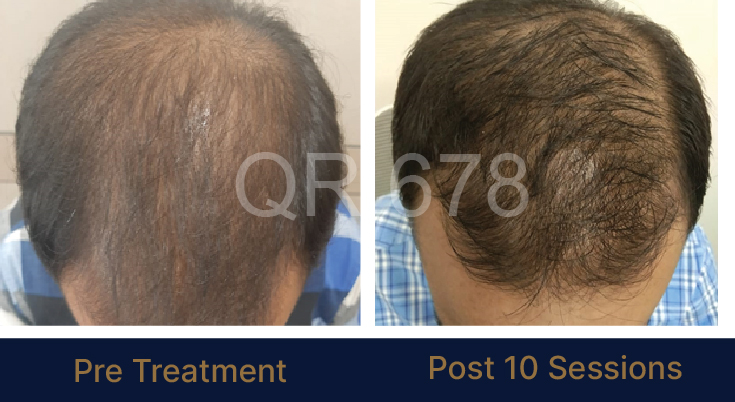
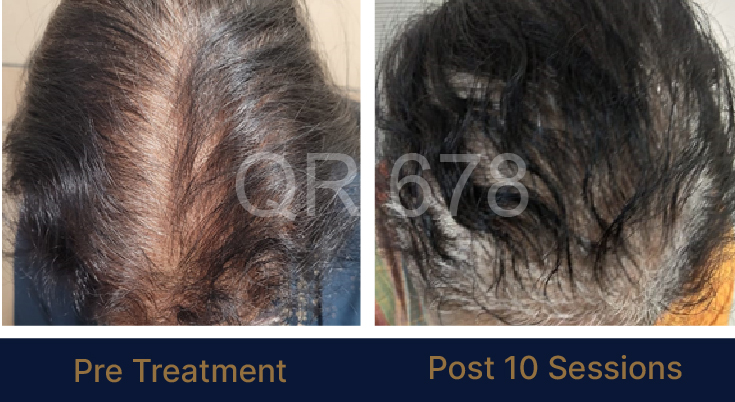
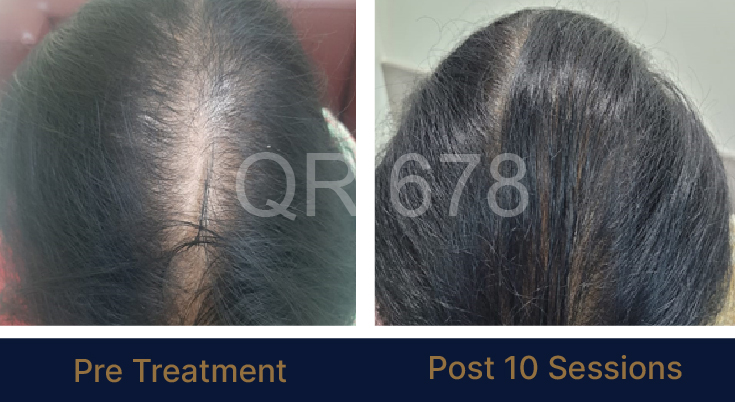
LIFE CHANGING RESULTS
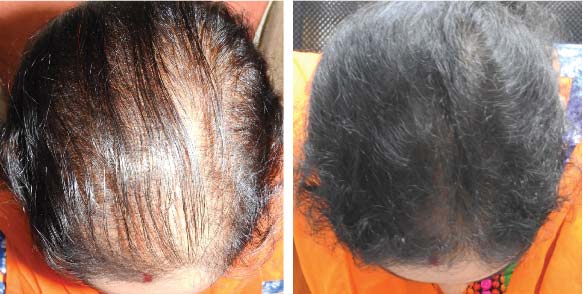
PRE TREATMENT
POST 6 SESSIONS
PRE TREATMENT
POST 6 SESSIONS

PRE TREATMENT
POST 6 SESSIONS
25 year old female from alopecia areata, was also diagnosed with hypothyroid. She achieved impressive results with QR678, a completely steroid-off treatment.

PRE TREATMENT
POST 6 SESSIONS
39 year old male suffering from male pattern hair loss for 15 years gained a significant amount of hair with QR678
MEDIA INTERVIEWS
TESTIMONIALS
HIGHLIGHTS IN MEDICAL CONFERENCES





CUTICON – December 2023
Dazzling moments at Cuticon 2023, Gujarat! QR678 took centre stage, earning accolades from top medical professionals.





QR678 made it’s presence felt at FADS (Face Aesthetics Dermatologists Society), the 7th International Conference held at Goa. Renowned dermatologists got QR678 Neo administered and shared their positive experience.




QR678® drew attention with an enlightening session and live demonstration at CDCON 2022 – a platform to challenge changing practices along the lines of Exploration and Innovation.
GLOBAL PRESENCE

OUR PARTNERS

















FAQ'S
How is QR678® better than other hair fall treatments?
QR678® is a mixture of natural growth factors that are already present in the scalp and therefore it is completely safe to use on everybody. It has the right concentration of specific hair growth factors and QR678® does not have any side effects, is performed as an outpatient procedure and the results are visible within eight weeks of starting the treatment. You can listen for yourself what the patients say about QR678® hair fall treatment https://www.youtube.com/watch?v=pIvjpQgSsrc
How often do I need to repeat QR678® treatment?
QR678® injections are administered in 8-12 sessions in about 3-4 weeks apart. The results are visible after few weeks. Once you have achieved the desired results the injections need not be repeated unless the hair fall starts again. This is because QR678® make the hair follicles healthy. Get to know more about why QR678® is highly effective in treating hair fall and increasing hair growth. http://www.qr678.in/
What precautions should I take after QR678® treatment?
QR678® is a completely safe treatment without side effects and you can go about your daily routine after the sessions. The only care you need to take is to use a gentle shampoo not before the next day of the treatment and avoid scratching and rubbing the treating area. If you have any other problem then consult your dermatologist. https://www.youtube.com/watch?v=LNm2z3EGLxg
Can QR678® be used after hair transplant?
Yes, using QR678® after hair transplant produces very successful results. QR678® gives faster density and reduces the catagen loss of transplanted hair. The growth factors in QR678® Neo help in faster healing and activate the dormant follicles and thus
– Increase the viability of hair follicles
– Improve healing and repairing of tissue process
– Strengthen the inactive hair follicles and stimulate hair growth
– Improves the health of existing hair and they will become fuller and stronger
QR678® can be given in multiple treatment sessions after three to four weeks after successful hair transplant surgery. QR678® can also be used before the hair transplant surgery to reduce the area of transplant and improve the results.
How is QR678® better than PRP and stem cell hair fall treatment?
QR678® is a relatively new treatment for hair fall and hair regrowth but in the short time of its invention QR678® has proven to be more effective than PRP and stem cell treatment for reducing hair fall. QR678® produces longer lasting and better results and is completely safe and without any side effects. QR678® can help treat and rogenetic alopecia, female pattern hair loss, and hair loss because of chemotherapy, seborrheic dermatitis, and alopecia areata.
PRP and stem cell on the other hand do not have the specific concentration of required growth factors, they lack standardisation in terms of treatment protocols and preparation of the solution injected and lack good quality studies to prove their benefits. produce guaranteed results and can cause tightness and tenderness in scalp,
scar tissue formation, calcification of the injected points, and formation of scar tissues. Both processes are still under investigation and their results are still awaiting confirmation. Dr Rinky Kapoor of The Esthetic Clinics explains the difference between QR678® and stem cell here: https://www.youtube.com/watch?v=wgNihm8ciu4 and
Can genetic hair loss be treated?
Yes genetic hair loss can be treated if the treatment is started in time. Heredity hair loss usually runs in the family and can affect both men and women. If you too have a close elder relative who lost his or hair in a patterned baldness then you should keep a close eye on your hair too. Hereditary hair loss usually happens in a pattern; M or W shaped in men and along the hair part in women. See the doctor immediately if you suffer from hair loss. There are many common and successful treatments available for genetic hair loss which include
– Topical and oral medications: These include Minoxidil topical lotion and Finasteride tablets.
– Hair transplantation using FUT and FUE methods.
– Nutritional supplements: Your physician might also suggest some hair supplements that can help arrest hair fall. Dietary supplements containing Vitamin A, C, and Biotin, zinc, and iron can also help control hair fall caused because of genetics.
Treatment option for Chemotherapy induced Alopecia?
Alopecia post chemotherapy is a very common consequence of cancer treatments and therapies and it is often the most psychologically and socially devastating side effect. Many patients shy away from the treatments because of fear of hair fall and hair loss. However, in most cases, such alopecia is reversible but the recovery takes several months and even up to a year. Some basic latest treatment methods will speed up the healing:
– Scalp Cooling: This is a FDA approved method for patients suffering from breast cancer. Scalp cooling or hypothermia results in constriction of the blood vessels in the scalp, which in turn leads to reduced delivery of chemotherapy to the hair. Scalp cooling devices consist of cooling unit that circulate the coolant on the scalp. This is done about 30 minutes before the start of chemotherapy. Patients can feel some mild discomfort, nausea, and dry skin because of scalp cooling.
– Topical Minoxidil: Minoxidil helps by increasing the blood flow to the hair follicle thereby preventing miniaturization of hair follicle.
– QR678® hair regrowth treatment: QR678® is a FDA approved, completely safe hair regrowth treatment that has no side effects or causes any discomfort to the patient. Clinical trials have proved QR678 to be effective in treatment of chemotherapy induced alopecia in both men and women. The results of QR678® last lifelong.